Solutions for influenza vaccines based on insect cell matrix

Insect cell matrix technology


In the global public health field, vaccine production technology is undergoing a quiet revolution, and insect cell matrix technology is at the forefront of this transformation.Seasonal influenza causes approximately [number missing] deaths worldwide each year.3 million to 5 million severe cases,290,000 to 650,000 deathsIts threat to public health should not be underestimated. Traditional vaccine production mainly relies on chicken embryo systems, which suffer from problems such as delayed virus strain matching, long production cycles (more than 6 months), the risk of egg allergies, and limited production capacity. The insect cell-baculovirus expression system (BEVS), as an emerging technology platform, provides a novel solution to these challenges.


1. Advantages of Insect Cell Matrix Influenza Vaccine
Insect cell matrix influenza vaccine technology adoptsInsect cell-baculovirusExpression System (BEVS)Viral structural proteins are expressed and self-assemble into virus-like particles without genetic material.(VLPThese VLPs can mimic the conformation of natural viruses, simultaneously activating humoral immunity (neutralizing antibodies) and cellular immunity, with higher safety. Compared to traditional chicken embryo production technology, insect cell platforms have multiple advantages:
High production efficiencyProduction cycles can be shortened from 6 months to within 4 months, which is especially important during a pandemic;
Avoid antigen mutationBecause it does not rely on chicken embryos for production, it avoids the adaptive mutation (antigen drift) of the virus on chicken embryos, such as the problem of insufficient protection rate of H3N2 vaccine in 2017-2018 due to antigen drift.
Better immunogenicityStudies have shown that VLP vaccines produced from insect cells can fully stimulate a highly efficient antibody response against influenza A/B viruses, and their stability is significantly better than that of traditional vaccines.
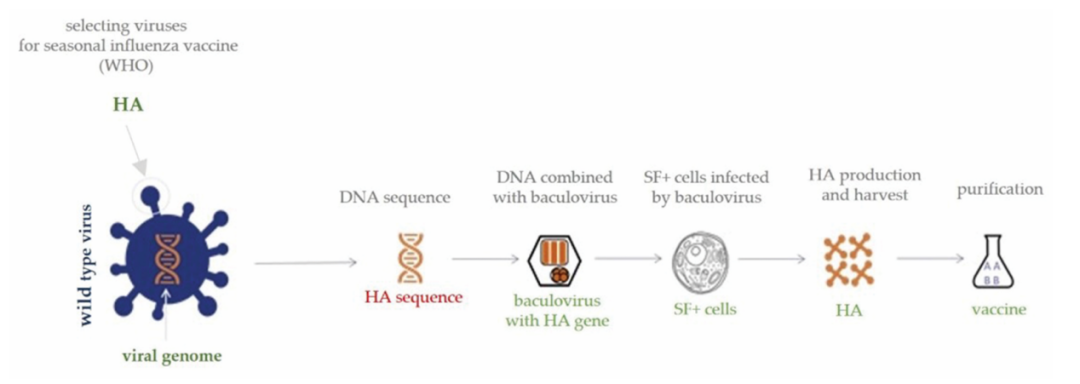
Figure 1: BEVS-based recombinant influenza vaccine (RIV) production process


2. Yishengke provides a comprehensive solution for insect cell matrix influenza vaccines.
2.1 SF9 cell rhabdovirus purification and H5 cell novavirus purification services
Insect cell-baculovirus expression system (BEVS)It is now widely usedDrug development, vaccine production, recombinant viral insecticidesAnd many other fields. And approximately [number missing] have already been established.More than 400 kindsContinuous insect cell lines exist, but only a few are used for commercial production. The earliest and most widely used insect cell line was...SF21SF21-derived cell linesSF9,It exhibits higher tolerance to osmotic pressure, pH, and shear stress, making it the most widely used cell line for expressing recombinant proteins.Another important cell lineBTI-TN-5B1-4It has a higher growth rate and protein production rate, and is namedHigh Five(H5)。
The Sf-9 cell line can rapidly amplify baculovirus, while the H5 cell line can produce more recombinant proteins and has more complex glycosylation modifications than Sf-9 cells.However, researchers have discovered that SF9 cell lines harbor [a latent [problem]].SF (specimen)(sf-rhabdovirus, sf-rv); latent in the Hi5 cell lineGiant freshwater shrimp nodavirus(Macrobrachium rosenbergii Nodavirus, MR-NV),This has caused concern among people.Although there are currently no literature reports that the above viruses pose a threat to humans or mammals, in order to ensure the absolute safety of biological products, regulatory authorities (as shown in the red box in Figure 2) and the industry generally require the elimination of species-specific viruses.
Yishengke can provide SF9 cell rhabdovirus purification and H5 cell novavirus purification services.Figure 3 shows the screening process for virus purification, with a screening cycle of 6-9 months.Currently, Yishengke has completed insect cell virus removal and purification services for more than 5 companies, and the fastest project has advanced to the Phase I clinical trial stage.
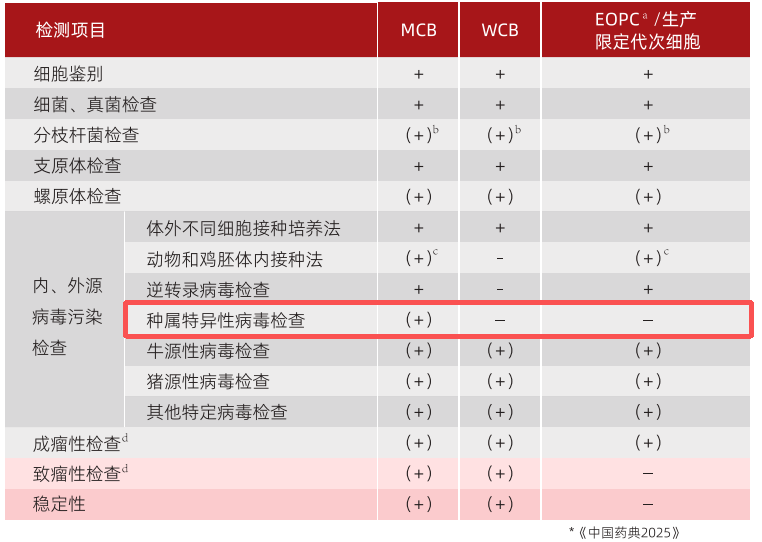
Figure 2: Requirements for cell assay items as specified in the pharmacopoeia
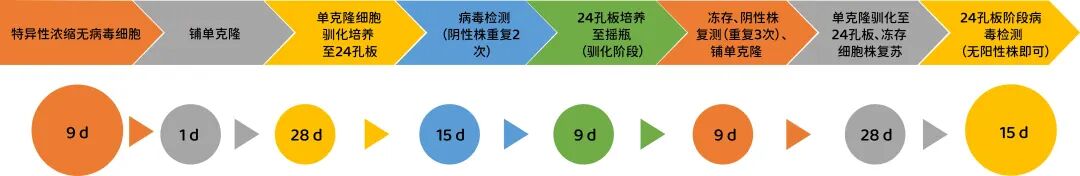
Figure 3: Insect cell purification and screening process
2.2 Insect cell-baculovirus vector construction and optimization service
The construction process of recombinant baculovirus is shown in Figure 4.Initially, recombinant baculoviruses were constructed by creating transfer vectors. Specifically, the polh gene and its boundary region were cloned into a bacterial plasmid, the coding region was deleted, and a GOI gene was inserted. This was then co-transfected with wild-type baculoviruses into insect cells. Homologous recombination resulted in polh being replaced by GOI, leading to the expression of the GOI protein and preventing the formation of polyhedrosis proteins. Distinction was achieved by comparing wild-type viral plaques with recombinant viral plaques. However, due to low recombination efficiency and insignificant differences, [further research was conducted].The following two improvement strategies are adopted.The most commonly used isBac-to-Bac systemThis system utilizes the baculovirus shuttle vector Bacmid, a baculovirus genome containing the E. coli replication origin, kanamycin resistance gene, Tn7 transposon, and transposition site attTn7. It can infect insect cells and replicate in E. coli. E. coli containing Bacmid is transformed with a transfer vector containing GOI located in Tn7. Transposition occurs via a helper plasmid, allowing the foreign gene to transpose into Bacmid. After blue-white screening, insect cells are transfected with Bacmid containing GOI to obtain recombinant baculovirus. Plaque screening of insect cells is not required.
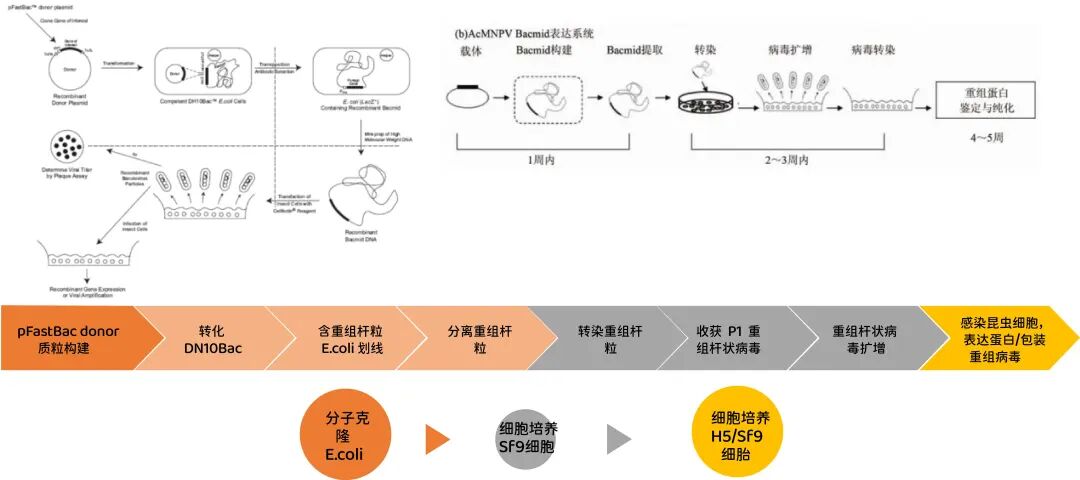
Figure 4: Construction and expression of baculovirus expression vector.
Although many proteins have been successfully expressed using baculovirus technology, problems remain due to cytopathic effects, the unpredictability of co-infection and co-expression, and the instability of the baculovirus genome.Some proteins still cannot be successfully expressed.As shown in Table 1,Yishengke can provide optimization of baculovirus expression systems, gaining a deep understanding of viral gene expression regulation and promoters.The selection principles (such as those of viral, synthetic, and insect-derived promoters) are used to leverage these complex gene regulatory mechanisms to increase the success rate of BEVS heterologous gene expression.
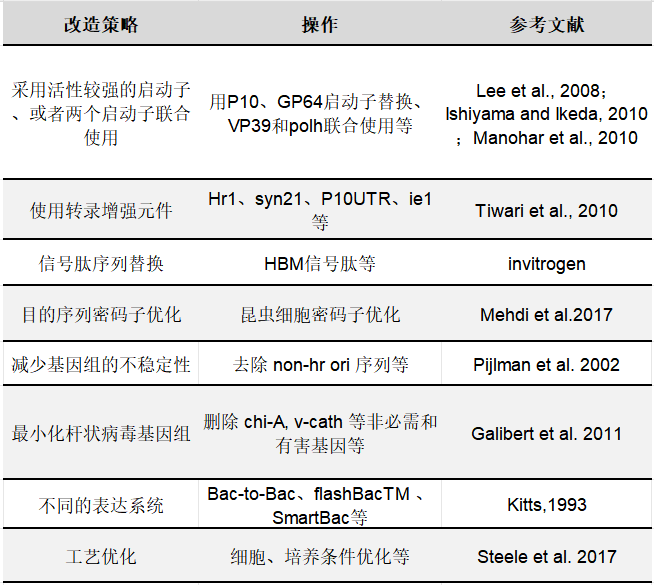
Table 1: Optimization Strategies for Baculovirus Expression Systems
2.3 Provide high-quality insect culture media andSubunit vaccinesPurification medium
Yishengke's independently developed Insect Cell Culture Medium IB907D has officially completed the DMF Type II filing with the US FDA.(Recordation No.: 040750). This achievement signifies that its industrialization level and quality assurance system have passed stringent international drug regulatory assessments, officially placing it among the leading players in the international biopharmaceutical core supply chain.IB907d is a serum-free cell culture medium, the latest universal serum-free and animal-free fully suspended culture medium for insect cells independently developed by Yishengke.Widely used inSf9, High Five cells, and other lepidopteran insect cellsSuspension culture and baculovirus system protein expression.
This product has four main features:
It exhibits long-term stable propagation, high-density growth, and a survival rate maintained above 95%.
Faster growth rate, higher density, and longer duration of maintenance;
Using Sf9 cells to amplify the virus resulted in a higher viral titer.
ExcellentBatch consistency(RSD<5%)。
In addition to IB907d, Yishengke also offers IB905 and IB907 insect cell suspension media, which are two universal serum-free suspension media for insect cells and are widely used in...BaculovirusA platform for expression.
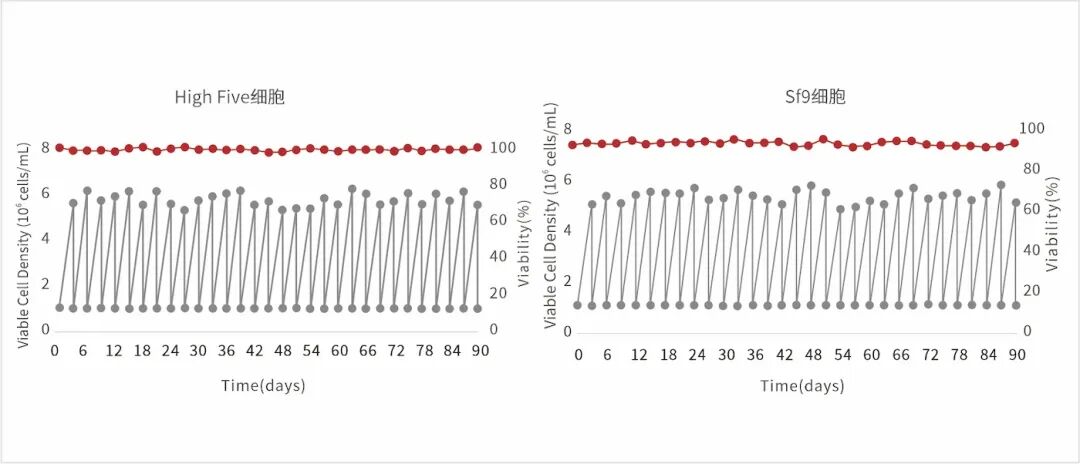
Figure 5: Stable passage of Hi5 and SF9 cells in IB907 and IB907d culture media, respectively.
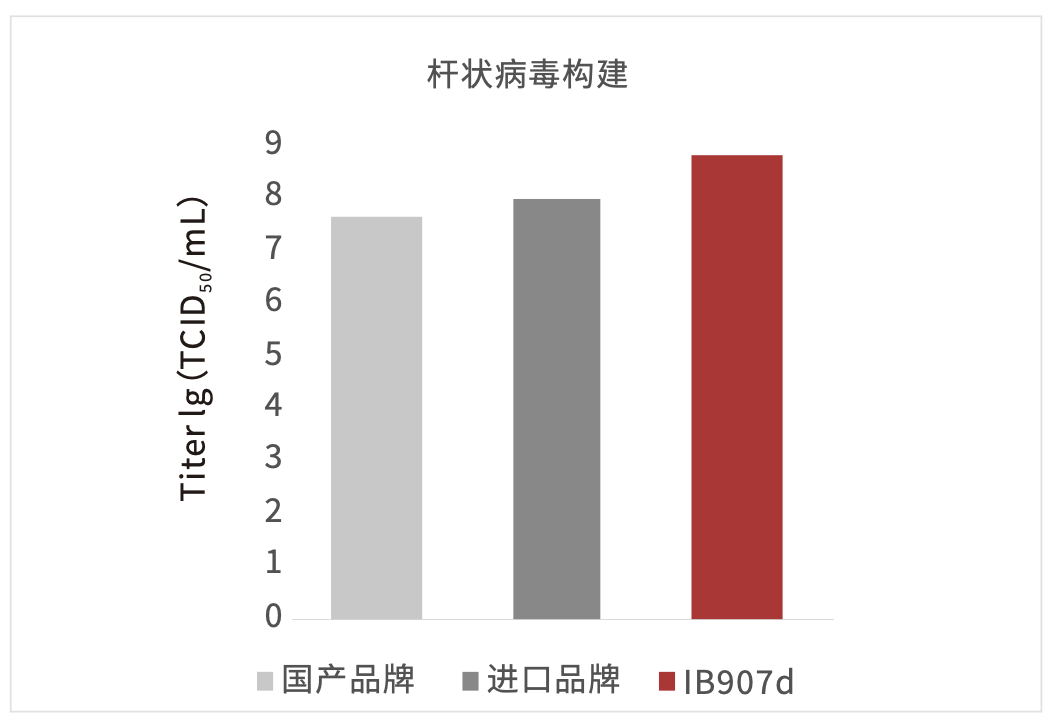
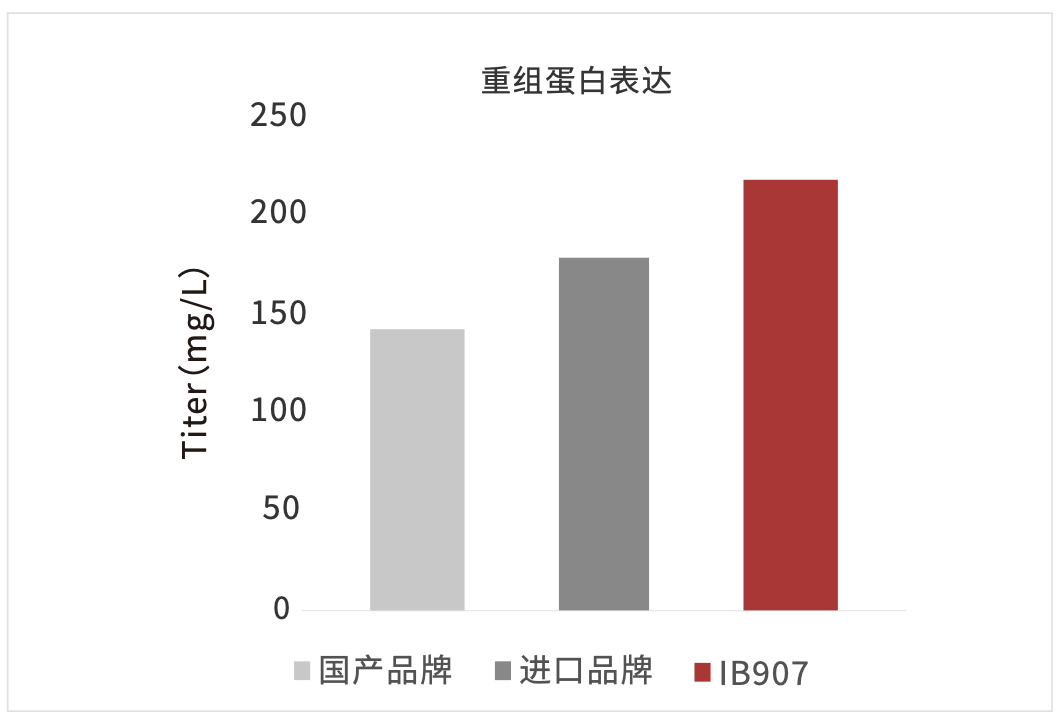
Figure 6: SF9 cells amplifying high-titer virus in IB907d (left)
Figure 7: Hi5 cells are highly expressing the protein in IB907 (right).
Yishengke has built GMP-compliant culture medium production workshops and purification packing material production workshops.Protein purification media products includeAffinity Chromatography、Ion exchange chromatography、Gel filtration chromatography、Hydrophobic ChromatographyFour main product categories. These provide high-quality purification media for recurrent influenza subunit vaccines expressed by the Kundalini system, ensuring the safety and regulatory compliance of the final product.
Gel filter mediaThis step removes residual host proteins, nucleic acids, or aggregates, while simultaneously replacing the buffer solution. It offers high resolution but low throughput and is typically used as the final purification step.
Multimode TomographyFor example, Aplo Shell series resins combine ion exchange and molecular sieve effects to remove multiple impurities in one step;
Ion exchange chromatography (IEX)Based on the surface charge characteristics of VLPs, anion exchange (such as Q column) or cation exchange (such as SP column) should be selected, and pH and salt gradient should be optimized to avoid particle aggregation.
Hydrophobic interaction chromatography (HIC)Based on the hydrophobicity of VLP surfaces, it is suitable for some non-coated VLPs.


3. Application Prospects and Outlook
along withWHO recommends quadrivalent vaccine(Covering two types of influenza A and two types of influenza B viruses), the global vaccine industry urgently needs an efficient, rapid, and safe production platform. In 2022, the quadrivalent influenza vaccine market reached $5.8 billion, with a compound annual growth rate exceeding 8%.There are already successful precedents for the application of insect cell technology in vaccine production.In 2009, Protein Sciences, Inc. in the United States produced the first batch of approximately 100,000 doses of H1N1 influenza vaccine using insect cell technology. Significant progress has been made in recent years. In 2024, scientists in Taiwan developed a quadrivalent virus-like particle (VLP) vaccine based on the insect cell baculovirus expression system (BEVS).Studies have shown that this vaccine can fully stimulate a highly efficient antibody response against influenza A/B viruses, and its stability is significantly better than that of traditional vaccines.
Zhejiang Yishengke Biotechnology Co., Ltd.'s insect cell culture medium IB907d has completed the DMF Type II filing with the US FDA, which means that drugs produced using its culture medium will have a simpler application process and faster approval speed.
Quadrivalent VLP vaccines produced using insect cell technology canFully stimulateHighly effective antibody responses against different types of influenza viruses, plusYishengke provides virus purification and screening services and high-quality culture media.This provides a powerful solution for dealing with the influenza epidemic.
In the future, with the continuous maturation of technology and the improvement of regulatory policies, insect cell matrix influenza vaccines are expected to be more widely used globally and become an important tool for protecting public health.
 +86 (0575) 8070 9355
+86 (0575) 8070 9355
 +86 (0575) 8070 9355
+86 (0575) 8070 9355
 marketing@yskbio.com
marketing@yskbio.com
 No. 16, Juli Road, Zhuji City, Zhejiang Province
No. 16, Juli Road, Zhuji City, Zhejiang Province
©2023 版权所有
