Common problems and solutions for human pluripotent stem cell (hPSC) culture

Science and Technology
hPSC culture
01
Poor cell adhesion/high cell mortality
Possible reasons:
Insufficient or inactivated matrix (Matrigel, Laminin, Vitronectin, etc.)
The passage method was too harsh, causing severe cell damage.
ROCK inhibitors not used or withdrawn too early
Solution approach:
Ensure proper substrate dilution and plating, and avoid repeated freeze-thaw cycles.
Recommended useEDTAGentle digestion avoids complete single-cell formation, as enzymatic digestion causes significant cell damage and can easily lead to differentiation in the long run.
Generally, a ROCK inhibitor is added during resuscitation to help cells adhere on the first day. Subsequent medium changes do not require the addition of the inhibitor. However, if poor cell adhesion persists, you can try adding the ROCK inhibitor to the culture medium again.
Healthy hPSCs have a high nucleus-to-cytoplasm ratio, prominent nucleoli, multiple nucleoli, and can form compact clonal clusters. The cells are flat and dense, with clear and smooth edges. The image below shows H9 cells cultured in Stem-Ex hPSC maintenance medium.
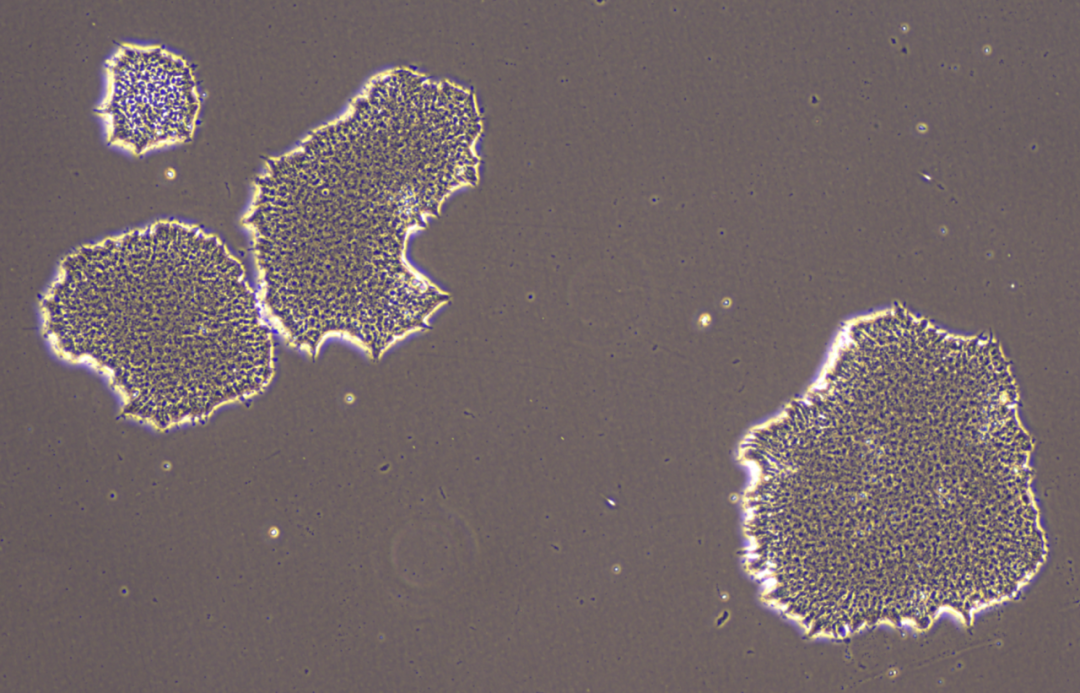
02
cellExcessive differentiation
Possible reasons:
Culture medium failure or uneven composition
Too low or too high a culture density leads to an imbalance in intercellular signaling.
Excessive passage intervals lead to excessive cell fusion.
Solution:
Ensure the culture medium is fresh, no more than 2 weeks old, and frozen components should not be repeatedly frozen and thawed.
Appropriately control the vaccination density (generally, 20-40% confluence is optimal).
Passage every 3–5 days to avoid excessively long culture times.
Common pluripotency indicators include SSEA4 and Tra-1-60, which are used in Stem-Ex hPSC maintenance medium.In the middle, after multiple generations of culture, the pluripotency index of hPSC exceeded 99%.
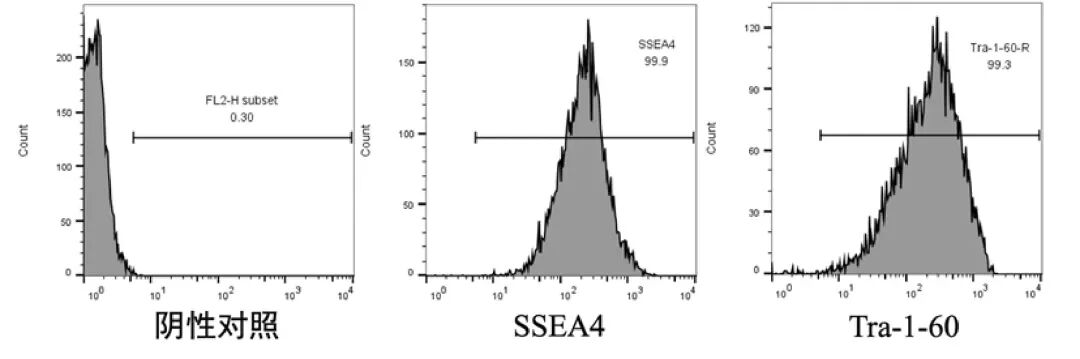
03
thinabnormal cell morphology
Performance:
Cells become elongated and flattened, losing their typical round/tight colonies.
Cells at the periphery of the colony disperse, showing a clear trend of differentiation.
Possible reasons:
Uneven substrate concentration and incomplete substrate spreading
Excessive cell tension can induce differentiation.
Culture medium contaminated or with unstable composition
Solution approach:
Check if the substrate coating is uniform
Confirm batch stability of culture medium
Investigate for contamination: bacteria, fungi, mycoplasma
Resuscitate cell culture
04
Mycoplasma contamination
Common symptoms:
Slow cell growth and abnormal morphology
Abnormal pH or turbidity in the culture medium
Solution:
Do it regularlyMycoplasma testing(qPCR, fluorescent dyes)
Cell lines must be discarded in cases of severe contamination to prevent cross-infection.
05
freezeSome solutions for low survival rates after survival/resuscitation
cryopreservation process:
cryopreservationLogarithmic growth phaseCells are generally available 2-4 days after passage, slightly earlier than the passage time.
Pay attention to digestion procedures; the diameter of cryopreserved cell clusters should be 50-200 micrometers.
Pay attention to the freezing procedure; if using a cryovial, do not store it at -80 degrees Celsius for too long.
recovery process:
Use the same culture system as before freezing, including the culture medium and coating matrix. If you need to change the system, you can try a gradient change during the second generation after thawing.
For the first generation after resuscitation, a ROCK inhibitor should be added to the culture medium.
During resuspension, cells are first added to centrifuge tubes, and culture medium is added dropwise to the cell pellet.
 +86 (0575) 8070 9355
+86 (0575) 8070 9355
 +86 (0575) 8070 9355
+86 (0575) 8070 9355
 marketing@yskbio.com
marketing@yskbio.com
 No. 16, Juli Road, Zhuji City, Zhejiang Province
No. 16, Juli Road, Zhuji City, Zhejiang Province
©2023 版权所有
