MDCK cell-based influenza vaccine solutions
Science and Technology
MDCK cells
Influenza as aSeasonal epidemicsIt causes a large number of infections and deaths worldwide every year.Traditional chicken embryo matrix vaccines have many limitations, while influenza vaccine production technology based on MDCK (Madin-Darby Canine Kidney) cells offers an innovative solution to these problems.This article will detail the overall solution for MDCK cell matrix influenza vaccine and introduce the capabilities of Zhejiang Yishengke Biotechnology Co., Ltd.Low tumorigenicity/non-tumorigenicityMDCK cell screening services and specialized culture media.
01
Advantages of MDCK cell matrix vaccine
Traditional chicken embryo cultured influenza vaccines have significant limitations: viral antigenic sites are prone to adaptive mutations, reducing vaccine effectiveness; production capacity is limited by chicken embryo supply, making it difficult to meet global demand during a pandemic; and they may also cause adverse reactions in people allergic to egg protein. In contrast,MDCK cell matrix influenza vaccine has significant advantages:
Higher antigen fidelity:To avoid adaptive mutations of the virus in chicken embryos, it can better match the circulating strains;
Production efficiency improvementEasy to scale up and not limited by chicken embryo supply;
Better securityNo egg white residue, reducing the risk of allergies;
Enhanced immunogenicityViruses cultured in cell matrix have more complex glycosylation patterns and can induce stronger cellular immune responses.
02
Zhejiang Yishengke can provide low tumorigenicity/non-tumorigenicity
MDCK Cell Screening Service
Zhejiang Yishengke Biotechnology Co., Ltd., located in Zhuji City, Zhejiang Province, has GMP-compliant culture medium production workshops and purification packing material production workshops, dedicated to providing high-quality consumables and technical support to the biopharmaceutical industry. Serum-free suspension cultured MDCK cells exhibit extremely high tumorigenicity; just 10 cells can induce tumor formation in nude mice. The National Institutes for Food and Drug Control (NIFDC) requires cell identification testing of MCB and EOPC for tumorigenicity/tumor-causing activity, as shown in Figure 1.Yishengke can provide services such as the complete suspension acclimation of adherent cells and screening for low-tumorigenic/non-tumorigenic MDCK cell lines.。
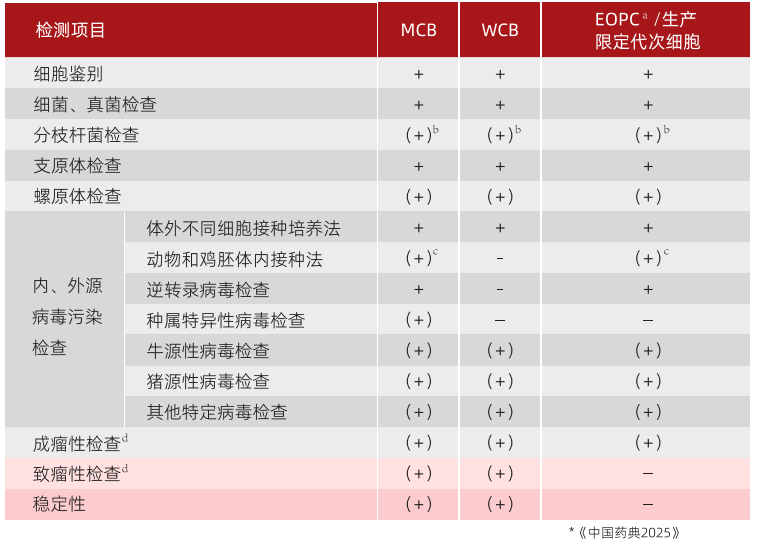
Figure 1. Requirements for Cell Testing Items
part.1
Serum-free suspension acclimation of adherent MDCK cells
Rapid cell suspension domestication strategy: Rapidly obtain a low-passage serum-free suspension growth population of MDCK cells, as shown in Figure 2.
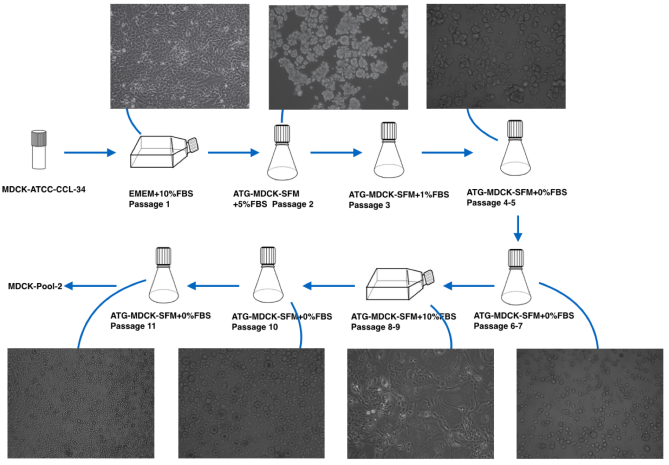
Figure 2. Rapid MDCK cell suspension domestication strategy
part.2
Screening of MDCK single-cell subclones using limiting dilution method
Yishengke obtained single-cell subclones of MDCK using the limiting dilution method, and then performed systematic evaluations on each cell line. The screening process evaluation mainly included:growth capacity;Extreme environmental toleranceDiameter distribution;Blood coagulation titer testTo assess the susceptibility of cells to influenza virus;Receptor abundance detection: Determine the virus's adsorption capacity;Tumorigenicity/tumorogenicity testingSafety is assessed through in vitro and in vivo testing.
Success stories
The research team at Yishengke obtained 108 single-cell subclones through screening. After two rounds of screening, three candidate cell lines were obtained, and finally, a subclone named MDCK-3 was selected as the candidate cell line. This cell line exhibited the following characteristics: the mode of chromosome number was 78.Nucleotype stability;Free from bacterial, fungal, mycoplasma and exogenous contaminants;nude miceNegative tumorigenesis testPathological anatomy of the inoculation siteNo significant difference compared to the negative control groupNeither cell lysate nor cell DNA inoculated into nude mice induced tumors, indicating that...Non-carcinogenic;No metastatic tumors were found.,TPD50(Median tumorigenic dose) ≤3.98×103Live cells/nude mouse。
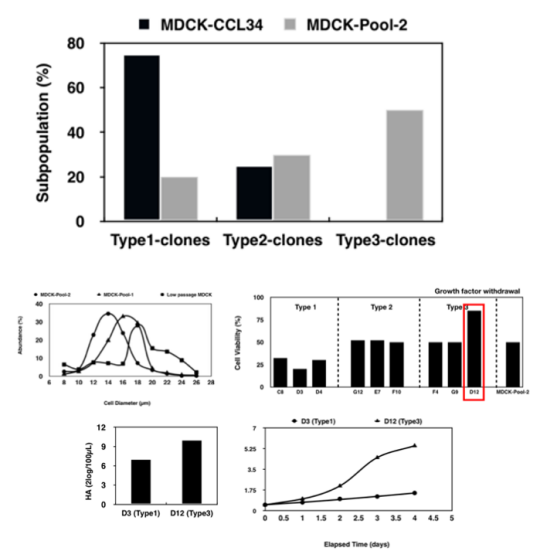
Figure 3. Comparison of diameter distribution, growth, and viral titer of MDCK-3 (Type 3) cells with other clones.
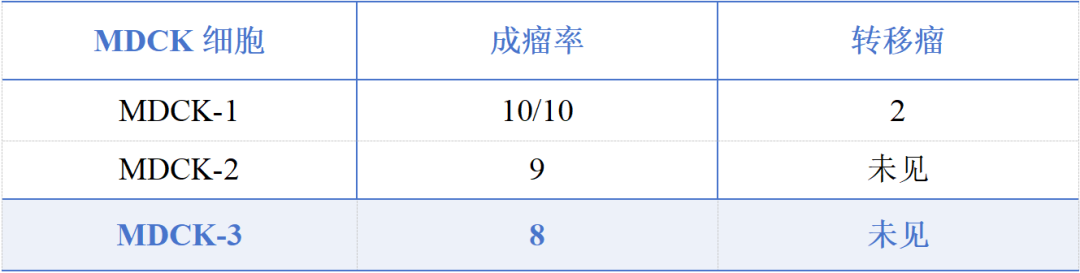
Table 1. MDCK Cell Bank Test Results - National Institutes for Food and Drug Control Report
03
Yishengke MDCK Special Cell Culture Medium
(Using MDCK-3 cells to verify the performance of the culture medium)
Zhejiang Yishengke has developed a TS specifically targeting MDCK cells.(True Suspension)Series of high-end culture media(Growth medium: MDCKS05 and virus amplification medium: MDCK307V), solving the technical difficulties of upgrading MDCK cells from adherent culture to full suspension culture.Features of Yishengke's TS series culture media:
Serum-free formulaReduce the risk of contamination from exogenous factors and improve product consistency;
Chemical composition limitationClearly defined components facilitate process standardization and regulatory approvals;
Support high-density growth(See Figure 4): It can achieve a cell density of tens of millions of cells per milliliter;
Stable cell growth after continuous passageCells can grow stably after 30 generations of culture, meeting the requirements for production generations.
(See Figure 5)
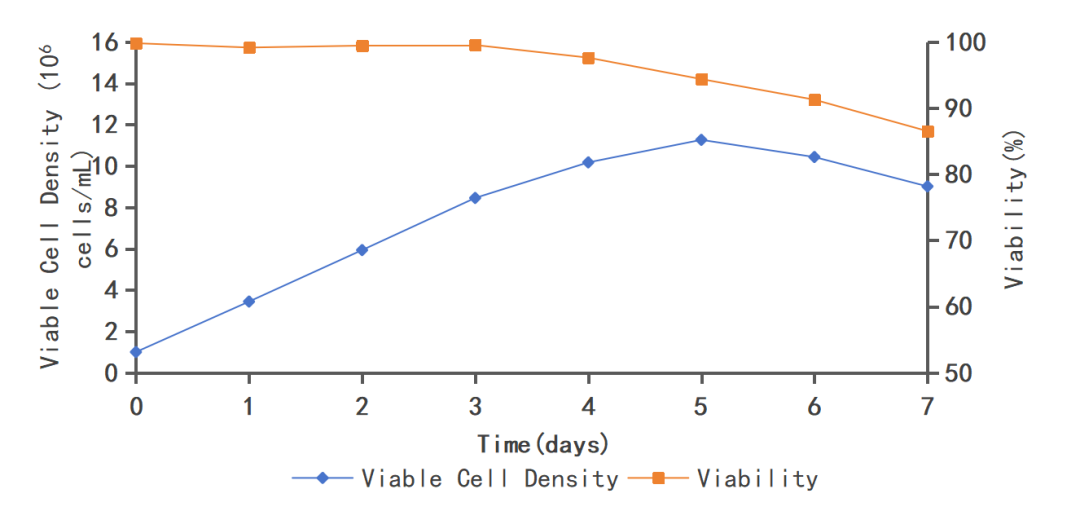
Figure 4. Batch culture of MDCK-3 in growth medium MDCKS05
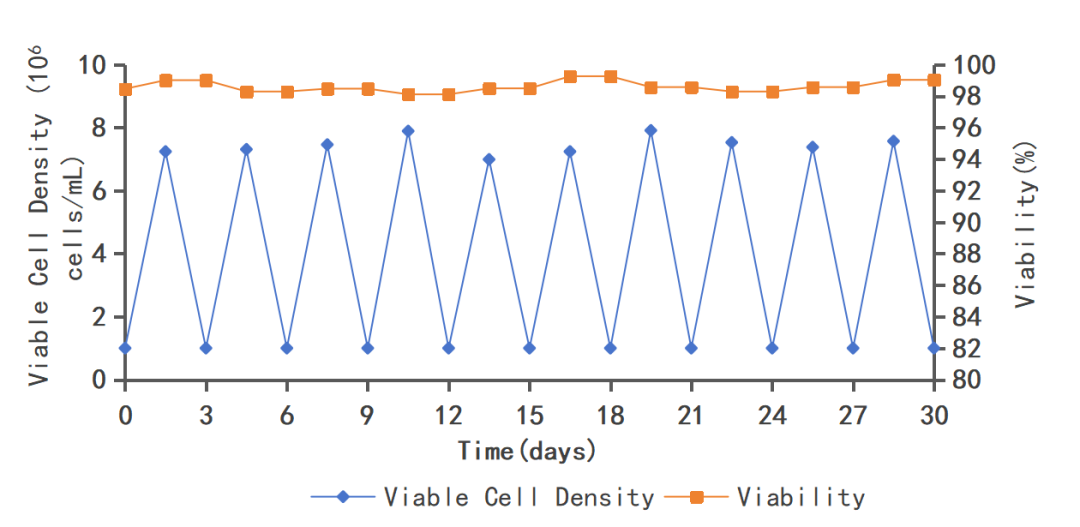
Figure 5. Successive passages of MDCK-3 in growth medium MDCKS05
Following the MDCK cell-based influenza virus production process outlined in Table 2, H1N1 and BV virus stock solutions were produced, and the TCID of the virus stock solutions was tested.50And HA (the results are shown in Figure 6).TCID of H1N1 and BV virus stock solutions produced by Yishengke New Technology50The results showed a significant improvement compared to the original process, while the HA results remained essentially unchanged. This indicates that the new technology from Yishengke did not cause viral drift or mutation during the process of increasing viral yield, thus maintaining its infectivity.。
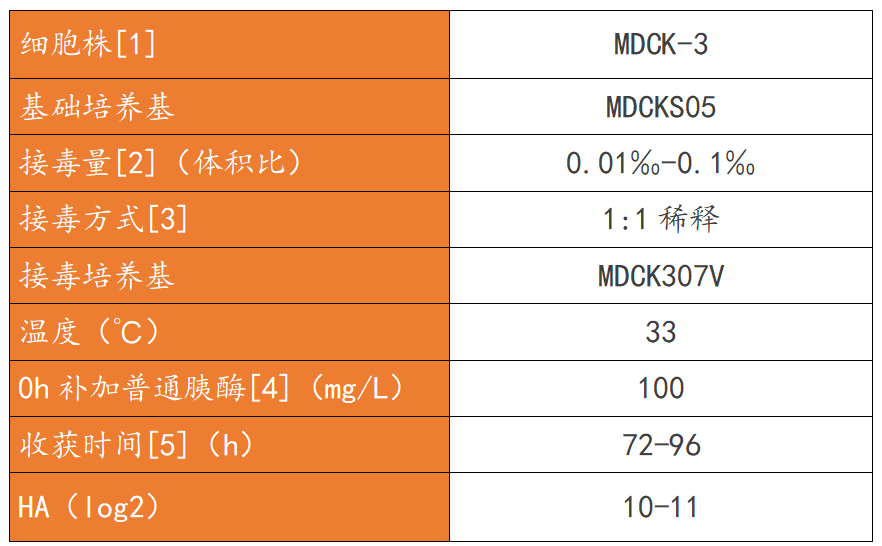
Table 2. MDCK Suspension Cell Influenza Transplantation Process
Notes:
[1] MDCK-3 cell culture conditions: temperature 37℃, rotation speed 140-160rpm, CO22Concentration 5%. Cell density ≥7×10⁻⁶ on day 3 of growth.6cells/mL, cell viability ≥95%.
[2] Since the inoculation amount varies among different strains, it is recommended that the inoculation amount be in the range of 0.01‰-0.1‰. An inoculation amount gradient can be designed to determine the optimal inoculation amount. The stability of the process is related to the quality of the seed virus. It is recommended to use high-quality seed virus [HA≥9(log2), virus content≥10].8.5(EID50/0.1mL)]。
[3] 1:1 dilution method: cell suspension after 72h of culture: fresh culture medium = 1:1.
[4] Normalpancreatic enzyme concentrationThe concentration is 100 mg/mL. For example, add 30 μL of ordinary pancreatic enzyme to a 30 mL infection system at a concentration of 100 mg/L.
[5] Harvest time: Due to the differences between different strains, the optimal harvest time needs to be determined based on the actual cytopathic effect. It is recommended that the harvest time be 72-96 hours.
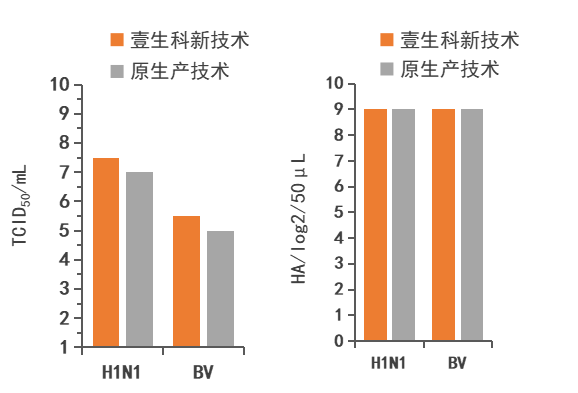
Figure 6. TCID of H1N1 and BV viral stock solutions50and HA test results
✦
•
✦
in additionIn addition to providing high-performance MDCK-specific culture media, Yishengke also offers comprehensive MDCK cell culture process optimization services.,include:thinCell passage parameter optimizationDetermine the optimal inoculation density;Optimization of cultivation conditionsDetermine the optimal rotation speed, incubation temperature, and carbon dioxide concentration;Virus production parameter optimizationDetermine the seed cell density, MOI, and TPCK-trypsin dosage. Simultaneously, it can also...Provide development, optimization, and validation of the MDCK cell matrix vaccine reactor production process.。
The main production process of influenza vaccine reactors based on MDCK cells includes the following steps: Reactor suspension cell culture stage: Optimized culture parameters in the bioreactor: pH: 7.0, dissolved oxygen: 50%, temperature: 37℃, stirring speed: 70-110 rpm/min (taking a 10 Leppdorf reactor as an example), deep aeration: 0.01~0.05 VVM; Virus inoculation and harvest optimization: The optimized virus proliferation process can significantly improve the hemagglutination titer of influenza virus; Virus replication is promoted by adding TPCK-trypsin; Cell-associated viruses are released using Triton-X100 cell lysis technology, further improving the hemagglutination titer.
04
Yishengke can provideSubunit vaccinesPurification packing
As a purification media supplier, Yishengke can provide media for the purification of subunit influenza vaccines. Our technical team can also provide efficient purification strategies to assist clients in developing or optimizing purification processes, accelerating product progress, and enabling them to enter the application or clinical trial stages as early as possible.Table 3 shows the results of the detection of the effective component HA and residual impurities after purifying the H1N1 virus stock solution using Yishengke purification packing material. All results met the limits required by the pharmacopoeia.

Table 3. Results of residual impurities detected during the purification of H1N1 virus stock solution
MDCK cell matrix influenza vaccine technology represents the future direction of influenza vaccine production.Zhejiang Yishengke Biotechnology Co., Ltd. has made significant contributions to the promotion and application of this technology by providing low-tumorigenic/non-tumorigenic MDCK cell screening services and specialized culture medium solutions.With the continuous maturation of technology and ongoing optimization of processes, MDCK cell-based influenza vaccines are expected to provide more effective, safe, and reliable protection for global influenza prevention and control.
 +86 (0575) 8070 9355
+86 (0575) 8070 9355
 +86 (0575) 8070 9355
+86 (0575) 8070 9355
 marketing@yskbio.com
marketing@yskbio.com
 No. 16, Juli Road, Zhuji City, Zhejiang Province
No. 16, Juli Road, Zhuji City, Zhejiang Province
©2023 版权所有
