Infectious Bursal Disease Virus Solution
I. Epidemiological Trends of Infectious Bursal Disease Virus
1.1 The emergence and continued prevalence of highly virulent strains
vvIBDV: Since its discovery in the late 1980s, highly virulent infectious bursal virus (IBDV) has become the dominant circulating strain globally. This type of strain can cause a mortality rate as high as 60%-70%, with more severe bursal hemorrhage and atrophy, and stronger immunosuppression.
Evolution of vvIBDV: Initially, vvIBDV was mainly concentrated in China, Japan, Southeast Asia, and Europe, but it is now widespread in major chicken-farming regions worldwide, and it continues to undergo genetic evolution, producing new sub-branches.
1.2 Emergence of new variants
This is one of the most worrying trends currently. The antigenicity of these strains has changed, enabling them to bypass the protection provided by traditional vaccines. Antigenic variants include: VarE and VarA in the US, and G2 in China. These strains: do not cause typical high mortality rates; clinical symptoms and lesions are not obvious and are easily overlooked. However, they lead to severe, long-term immunosuppression: flocks appear "healthy," but growth is stunted, vaccine responses are poor, secondary infections are common, resulting in significant economic losses. "Silent" outbreaks: due to the lack of typical symptoms, they are often only discovered through laboratory monitoring.
1.3 Expansion of host range and clinical manifestations
The age of onset has broadened: Traditionally, 3-6 weeks of age was considered the susceptible period, but now there are reports of cases in flocks as young as 10 days old and as late as the laying period. This has brought great difficulties to the development of immunization programs.
Atypical cases are increasing: Many infections present with subclinical symptoms, showing only decreased production performance and immunosuppression, without the typical hemorrhage and swelling of the infectious bursa of Fabricius.
1.4 An arms race between vaccines and wild-type viruses.
Viral evolution under immune pressure: On the one hand, IBDV is an RNA virus, which easily mutates during replication in animals or recombines with wild-type viruses in the field to produce recombinant viruses; on the other hand, to control vvIBDV, farms generally use live vaccines with moderate to strong virulence. This widespread immune pressure screens out variant strains that can evade vaccine protection. Risks of inappropriate vaccine selection: In areas where variant strains are prevalent, using only classic strain vaccines often results in inadequate protection, leading to immunization failure.
1.5 Mixed infections and environmental stress
Co-infection with other immunosuppressive diseases: IBDV frequently co-infects with other immunosuppressive pathogens such as avian infectious anemia virus, Marek's disease virus, and reovirus, producing a synergistic pathogenic effect that leads to far more severe consequences than single infections.
High viral load in the environment: Due to its extremely strong resistance, the virus often survives in the environment for extended periods. In chicken farms with continuous breeding over many years, the viral load accumulates continuously, leading to a persistently increased risk of early infection in chicks. Currently, the prevalence of infectious bursal virus (IBDV) exhibits the coexistence of multiple genotypes.
Therefore, when vaccinating farms, it is necessary to select vaccines containing circulating strains based on local epidemiological survey results to provide effective protection.
II. Existing Solutions
Vaccination is the core of IBD prevention and control, but the key lies in "precision" rather than indiscriminate vaccination.
2.1 Vaccine type selection
2.1.1 Live Vaccines: Mild: No damage to the bursa of Fabricius, but limited protection; mainly used in low-virulence areas or for basic immunization. Moderately Virulent: Currently the most widely used type globally, effectively resisting virulent attacks, but causing slight reversible damage to the bursa of Fabricius. Moderately Positive/Virulent: Used only in areas with high vvIBDV prevalence; strong protection, but high risk of bursal damage, requiring cautious use.
2.1.2 Inactivated Vaccines: Primarily used in breeder hens before laying eggs to stimulate the production of high and uniform antibody levels, which are then passed to chicks through eggs, providing robust maternal antibody protection.
2.1.3 Immune Complex Vaccines: Immune complex vaccines combine live virus with specific antibodies, overcoming maternal antibody interference and achieving early, uniform immunization. Virus-like Particle Vaccines: Safe, highly effective, and providing broader-spectrum protection; the future direction of development.
2.2 Key aspects of immunization programs: monitoring and timing
Vaccine selection considerations: Selection must be based on the prevalent strains in the farm or region. In areas where vvIBDV is prevalent, moderately virulent vaccines should be used. In areas where variant strains are prevalent, vaccines matching the antigens of the prevalent strain should be used (such as vaccines for specific serotypes or multivalent vaccines). Monitoring maternal antibodies: This is the cornerstone of developing an immunization program. Maternal antibody levels are detected using ELISA by regularly collecting chick serum. Goal: To identify the "window period" during which maternal antibodies decline to an insufficient level to provide protection, and to administer the first immunization within this window. Benefits: Avoids immunization too early (maternal antibodies neutralize the vaccine virus, leading to immunization failure) or too late (wild-virus infection has already occurred).
III. Yisheng Biotech provides solutions.
3.1 Using the Bac-to-Bac system to construct a baculovirus for screening and expression of IBDV protective antigens.
Duration: 1-2 months
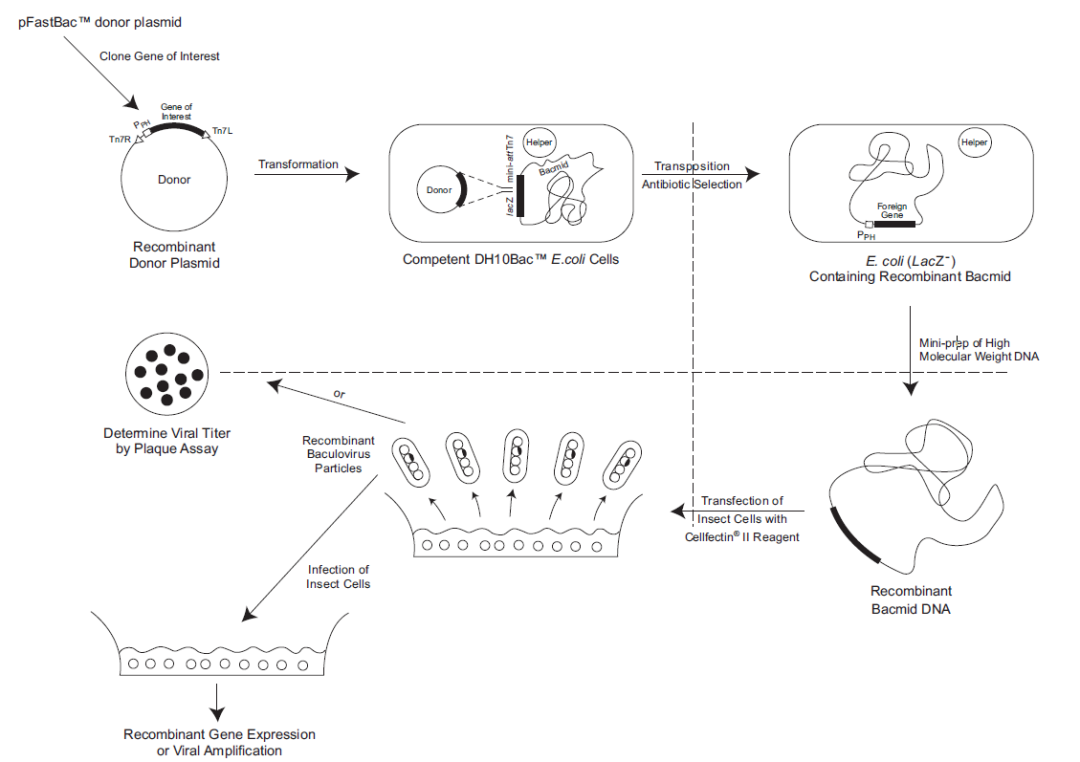
3.2 High-Expression Insect-Baculovirus Production Technology
Insect Cell Culture Medium
• Serum-free, fully suspended culture medium
• Stably maintains insect cell growth
• Widely used in baculovirus system protein expression in SF9 and High Five cells
• Supports high-density cell growth, efficient protein expression, and large-scale production scaling
Time required: 1 month
Long-term stable subculturing, high-density growth, and cell viability maintained at over 95%.
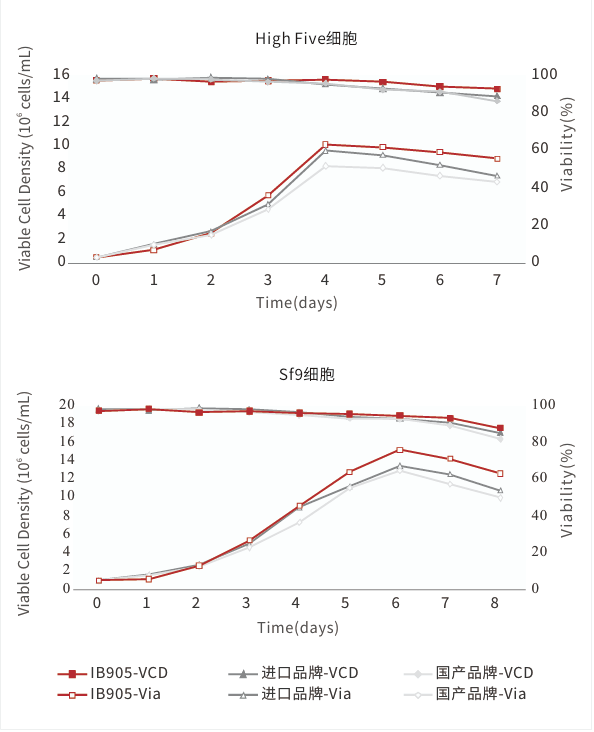
Faster growth rate, higher density, and longer duration.
3.3 Process development and scaling up
Expression verification: SDS-PAGE, ELISA, and other techniques were used to verify the correct expression and yield of the VP2 protein.
Culture condition optimization: Optimize cell culture conditions (pH, temperature, dissolved oxygen, and feeding strategy) in shake flasks and bioreactors to improve protein expression levels and cell density.
Large-scale suspension culture: Batch, fed-batch, or perfusion culture is performed in 50L-200L bioreactors to establish a stable and controllable upstream production process.
Further purification: The highly concentrated, purified VP2 protein is formulated with a suitable adjuvant to enhance the immune response.
用时:约1-2月。
3.4 Animal testing evaluation
Safety: This demonstrates that the vaccine is safe for the target animal (chickens) and does not cause significant adverse reactions.
Efficacy: This demonstrates that the vaccine can stimulate the body to produce sufficient immune protection, effectively preventing infectious bursal disease virus infection and related diseases.
Efficacy: To determine the minimum antigen dose required for the vaccine to produce a protective effect (minimum immunizing dose).
Timeline: 2-3 months.
In summary, the latest subtype of infectious bursal disease virus (IBDV) VP2 protein can be obtained in approximately 3 months; a stable insect cell line engineered to express high levels of VP2 protein can be obtained in approximately 6 months.
Table 1: Advantages and Disadvantages of Live Vaccines and Yisheng Bio's Insect Baculovirus Subunit Vaccines
Live vaccine | YSK Insect baculovirus subunit vaccine |
The immune response is comprehensive and potent. It not only stimulates humoral immunity (producing neutralizing antibodies) but also triggers a strong cellular immune response. Because the virus replicates in the body, the immune response is closer to that of a natural infection, providing protection that is usually more comprehensive and longer-lasting. | High safety and no risk of virulence reversion: Because it does not contain complete viral nucleic acid, there is no possibility of virulence recovery. Furthermore, chickens do not have baculovirus receptors, eliminating the possibility of recombination between the IBDV target gene fragment in the baculovirus and field wild-type viruses. Virus-like particles effectively stimulate an immune response without immunosuppression: They do not replicate in the bursa of Fabricius, thus preventing bursal damage and immunosuppression. No exogenous viral contamination: The production process does not involve chicken embryos or live cells, eliminating the possibility of exogenous pathogen contamination. |
Its effectiveness is undeniable, and its application history is long. After decades of field use, its immunogenicity and protective range (especially against classic and highly virulent strains) have been fully validated. It is currently the mainstay of IBD control globally, and its technical solutions are very mature. | It can overcome the interference of maternal antibodies, has a high antigen content, and can neutralize maternal antibodies, effectively breaking through the blockade of maternal antibodies. |
It is inexpensive and easy to adopt. The production process (mainly in chicken embryos) is relatively mature and stable, with low cost, making it very suitable for large-scale farms. | It can be used for differential diagnosis. Some genetically engineered vaccines (such as subunit vaccines and virus-like particle vaccines) contain only major antigen proteins such as VP2. Wild-type virus infection in the field produces antibodies against other proteins (such as VP1). ELISA kits can distinguish between vaccinated chickens and chickens infected with wild-type viruses, which is beneficial for disease eradication and monitoring. |
There are various routes of vaccination, including drinking water, nasal drops, and eye drops. The procedure is simple and highly effective for large-scale immunization. | Precise design allows for the creation of vaccines that better match the antigenic characteristics of currently circulating strains, achieving more precise protective effects. |
Disadvantages
| Disadvantages
|




